
Assistant Professor Eun Ji Chung, the Dr. Karl Jacob Jr. and Karl Jacob III Early-Career Chair. Image/Hugh Kretschmer
Heart disease and stroke are the world’s two most deadly diseases, causing over 15 million deaths in 2016 according to the World Health Organization. A key underlying factor in both of these global health crises is the common condition, atherosclerosis, or the build-up of fatty deposits, inflammation and plaque on the walls of blood vessels. By the age of 40, around half of us will have this condition, many without symptoms.
A new nanoparticle innovation from researchers in USC Viterbi’s Department of Biomedical Engineering may allow doctors to pinpoint when plaque becomes dangerous by detecting unstable calcifications that can trigger heart attacks and strokes.
The research — from Ph.D. student Deborah Chin under the supervision of Eun Ji Chung, the Dr. Karl Jacob Jr. and Karl Jacob III Early-Career Chair, in collaboration with Gregory Magee, assistant professor of clinical surgery from Keck School of Medicine of USC — was recently published in the Royal Society of Chemistry’s Journal of Materials Chemistry B on 25 September 2019.
When atherosclerosis occurs in coronary arteries, blockages due to plaque or calcification-induced ruptures can lead to a clot, cutting blood flow to the heart, which is the cause of most heart attacks. When the condition occurs in the vessels leading to the brain, it can cause a stroke.
“An artery doesn’t need to be 80 percent blocked to be dangerous. An artery with 45% blockage by plaques could be more rupture-prone,” Chung said. “Just because it’s a big plaque doesn’t necessarily mean it’s an unstable plaque.”
Chung said that when small calcium deposits, called microcalcifications, form within arterial plaques, the plaque can become rupture prone.
However, identifying whether blood vessel calcification is unstable and likely to rupture is particularly difficult using traditional CT and MRI scanning methods, or angiography, which has other risks.
“Angiography requires the use of catheters that are invasive and have inherent risks of tissue damage,” said Chin, the lead author. “CT scans on the other hand, involve ionizing radiation which can cause other detrimental effects to tissue.”
Chung said that the resolution limitations of traditional imaging offers doctors a “bird’s eye view” of larger-sized calcification, which may not necessarily be dangerous. “If the calcification is on the micro scale, it can be harder to pick out,” she said.
The research team developed a nanoparticle, known as a micelle, which attaches itself and lights up calcification to make it easier for smaller blockages that are prone to rupture to be seen during imaging.
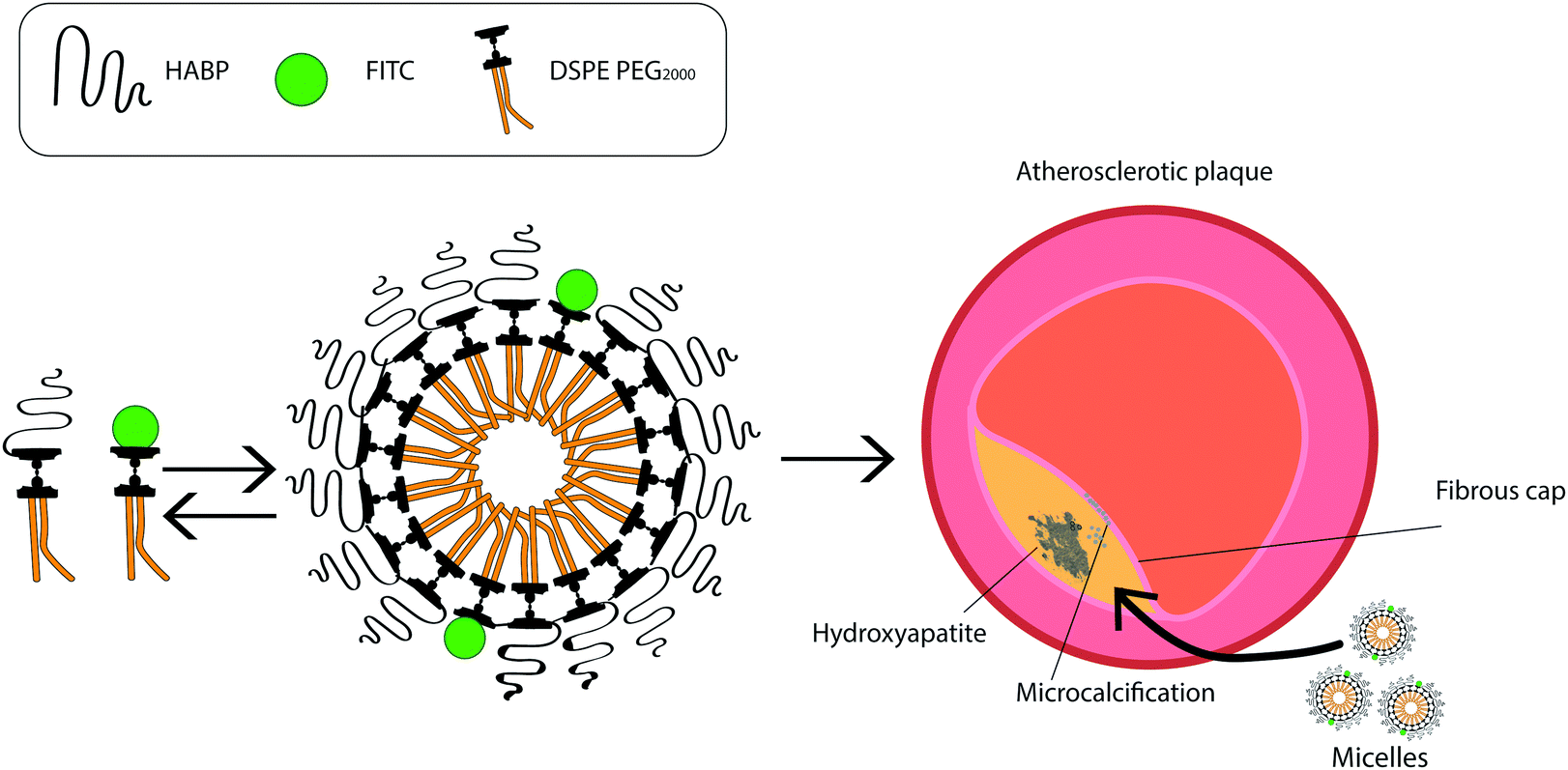
The Chung Lab team’s nanoparticle micelles (left) with fluorescent tags, attach themselves to microcalcification in atherosclerosis.
Chin said the micelles are able to specifically target hydroxyapatite, a unique form of calcium present in arteries and atherosclerotic plaques.
“Our micelle nanoparticles demonstrate minimal toxicity to cells and tissue and are highly specific to hydroxyapatite calcifications,” Chin said. “Thus, this minimizes the uncertainty in identifying harmful vascular calcifications.”
The team has tested their nanoparticle on calcified cells in a dish, within a mouse model of atherosclerosis, as well as using patient-derived artery samples provided by vascular surgeon, Magee, which shows their applicability not only in small animals but in human tissues.
“In our case, we demonstrated that our nanoparticle binds to calcification in the most commonly used mouse model for atherosclerosis and also works in calcified vascular tissue derived from patients,” Chin said.
Chung said that the next step for the team was to harness the micelle particles to be used in targeted drug therapy to treat calcification in arteries, rather than just as means of detecting the potential blockages.
“The idea behind nanoparticles and nanomedicine is that it can be a carrier like the Amazon carrier system, shuttling drugs right to a specific address or location in the body, and not to places that you don’t want it to go to,” Chung said.
“Hopefully that can allow for lower dosages, but high efficacy at the disease site without hurting normal cells and organ processes,” she said.
Published on December 5th, 2019
Last updated on February 8th, 2022













