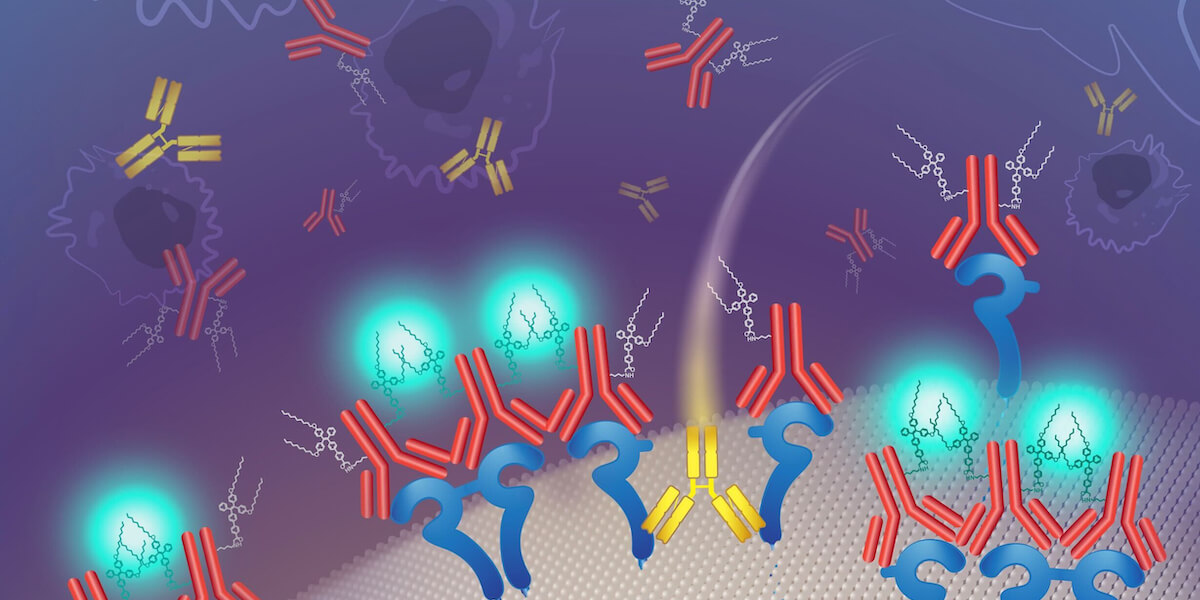
A conceptual drawing of a new fluorescent probe that illuminates protein clusters found in a subset of breast cancers (Illustration/Katya Kadyshevskaya)
As a young girl, Andrea Armani lost both grandmothers to breast cancer. Unfortunately, she is not alone.
Cancer continues to be the second most common cause of death in the US, after heart disease, according to American Cancer Society. About 1 in 8 women are expected to develop breast cancer sometime in their lives.
In 15% to 20% of breast cancers, there is a protein on the cell surface called HER2. This protein serves multiple roles in the fight against breast cancer – as a diagnostic indicator and as a therapeutic target.
The majority of diagnostics just look for the presence or absence of this protein. In contrast, therapeutic design strategies have taken two general paths: using the protein as an anchor site for known chemotherapeutics or disrupting the distribution of the protein on the cell surface. By forcing the individual HER2 proteins to be farther apart, the cell’s ability to function is modified. To pursue the second approach, it is necessary to both detect the presence of and measure the spatial distribution of HER2 on the cell.
“Most high throughput methods only tell you if HER2 is present or not. When developing therapeutics, this is only one half of a complex puzzle,” said Armani, the Ray Irani Chair in Chemical Engineering and Materials Science and a senior director at the Ellison Institute of Technology.
This protein organization is on the nanoscale. To see it, researchers use electron microscopy or super-resolution microscopy. While both methods are successful in detecting the arrangement, they are very time-consuming, because you can only image a small region of a sample at a single time. “Because you can only look at a little bit of a sample, you could miss something really important,” Armani said.

Andrea Armani (Photo/Courtesy of USC Viterbi)
Against this backdrop, Armani and Yasaman Moradi, one of her Ph.D. students in chemical engineering, have developed a light-emitting molecule that can detect changes in HER2 organization and clustering in response to therapeutics. Additionally, the material is compatible with conventional and automated imaging methods, like the automated high throughput imagers used in the pharmaceutical industry. That means “scientists can evaluate entire cell plates at once, up to 1,536 well plates or several thousand cells,” Armani said, instead of just one cell. The result: faster results at a lower cost.
This advance relies on a unique feature of their material. It only lights up when unable to freely move or rotate. To take advantage of this property, the USC researchers modified the material to only stick to HER2 proteins and added some long arms to increase molecule-molecule interactions. As a result, they can detect when the proteins are clustered because the light-emitting material becomes tangled and lights up.
“Because our imaging material provides information about what is happening on the cell surface and is compatible with high throughput methods, we can speed up the development of better therapeutics and have the opportunity to save more lives,” said Moradi, lead author of “Detecting Disruption of HER2 Membrane Protein Organization in Cell Membranes with Nanoscale Precision,” along with Armani and Dr. Jerry Lee, who holds a joint appointment at the Keck School of Medicine of USC and the USC Viterbi School of Engineering. Their research was just published in “ACS Sensors,” an American Chemical Society journal.
Armani said she believes the fluorescent probe holds the promise to increase researchers’ understanding of a variety of diseases beyond cancer, including neurological disorders like Alzheimer’s and epilepsy. “While this tool was demonstrated using HER2 as a model system, the fluorescent probe can be designed to study other biological systems by simply changing the targeting molecule,” she said.
The Office of Naval Research, Henry M. Jackson Foundation for the Advancement of Military Medicine and the Ellison Institute of Technology supported the research.
Published on November 14th, 2023
Last updated on November 17th, 2023











