
Newly hatched Hawaiian Bobtail Squid E. scolopes recruits V. fischeri to the ciliated surface of their nascent light-emitting organ from inhaled seawater. Photo/Janna Nawroth
Imagine a club scene—a bouncer at a velvet rope selects which individuals get into the club. This, explains Eva Kanso, a professor of mechanical engineering at USC Viterbi School of Engineering, is what cilia do in an organism. Kanso applied the analogy to explain her new paper, “Motile cilia create fluid-mechanical microhabitats for the active recruitment of the host microbiome,” co-authored with researchers from the Pacific Biosciences Research Center at the University of Hawaii at Manoa and from Stanford, to explain the active role that cilia have in ensuring certain bacteria are kept out of an organism while other symbiotic bacteria are selectively permitted to enter.

Visualization of 2 types of cilia-mediated flow that facilitate the capture and screening of bacterial symbionts (red) at host surface epithelia (green). Photo/Janna Nawroth
The paper, published in the Proceedings of the National Academy of Sciences, describes a framework for the role of fluid mechanics in letting symbiotic bacteria in an organism and enhancing chemical communication between the symbiont and the host organism. The results are contrary to previous research which assumes that cilia solely play a “clearance function.” They could shed light on the role cilia—which are the size of one hundredth of a single human hair—play in human respiratory system and even in the reproductive systems and the brain. Their findings could also provide insights on how cilia dysfunction within organs affect for example, pulmonary conditions or infertility (how cilia help sperm reach eggs).
To learn about how cilia might work in the human body, Kanso, in collaboration with symbiosis expert McFall-Ngai and biofluid expert Janna Nawroth, studied bobtail squid. The researchers examined how these squids in their nascent stage allow symbiotic bacteria Vibrio Fischeri to enter into their ciliated light organs, which play a crucial role in camouflaging the ink sacks of the otherwise translucent organism while they hunt for food at night. The scholars sought to know: why does this bacterium gain access and why do all bacteria fail to accumulate within the squid’s light organ? In addition, they sought to explain what, if any, is the role of cilia in allowing access?
Kanso and McFall-Ngai with lead authors Janna Nawroth, a principal investigator at Emulate Inc, and Hanliang Guo, a PhD student in Kanso’s lab at USC, put the squid under a microscope and then exposed it to water containing Vibrio Fischeri bacteria. The process mimicked what happens in nature: the bacteria ended up in the correct spot within the squids’ light organs. To determine if cilia were passive or active within this process, the researchers needed to understand if the bacteria swam into the ink sack or in fact were helped by other structures and processes within the squid host. The researchers then repeated the same experiment without live bacteria—but with particles of the same size as the bacteria, namely one micron in diameter. What they found was that the particles accumulated in the same spot –demonstrating that a physical mechanism in the host (the squid) was at play. The second phase of experiment then included larger particles (about 4 microns). One might have presumed that the larger particles would have a greater probability of contact with the light organ—but they didn’t. This indicated that direct interception was not the dominant mechanism for particle capture and that some other factor was at play. The researchers set out to examine the role of the fluid flow generated by the cilia in filtering particles.
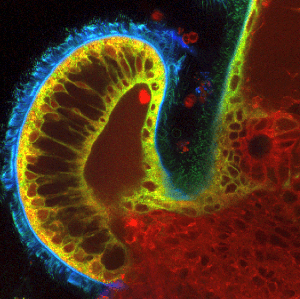
The squid’s internal light organ features several different populations of cilia (green/blue) that coordinate their beating activity to recruit symbiont bacteria from the seawater and facilitate their migration to the pores (right side), where they enter the organ for life-long colonization. Photo/Lab of Margaret McFall-Ngai
Upon further investigation, Kanso and Nawroth discovered that a vortical or “donut-like” flow generated by the cilia was kicking away most particles. The role of the fluid motion in filtering particles by size was verified using a physics-based mathematical model developed by Kanso and Guo. Kanso describes the role that cilia seemed to be playing as a “mechanical gate.”
The researchers then mapped out the ciliated surface of the whole light organ and the flow field it generates. One of the core findings was that there were two distinct flows taking place by two different types of cilia. Longer cilia move in a “wave-like” fashion which creates a vortical flow field that filters particles and then shorter cilia which beat randomly keep the particles in place and gently mix the local flow. This random motion by the cilia and fluid mixing enhance the chemical screening of bacteria. To further prove the important role played by cilia, the researchers also found that if cilia are “killed,” particles will accumulate everywhere in the organism.
Kanso and her collaborators are now developing a microfluidic platform to test the response of Vibrio Fischeri to distinct flow and chemical signals presented by the ciliated light organ of the host. This platform will be used as a research tool to investigate the relative importance of each of these signals in the recruitment of bacteria to ciliated surfaces.
Published on September 8th, 2017
Last updated on February 1st, 2018











