Illustration/Madelin Lum
In his 1971 State of the Union address, President Richard Nixon dedicated the country to finding a cure for cancer. “The time has come in America when the same kind of concentrated effort that split the atom and took man to the moon should be turned toward conquering this dreaded disease,” he said. “Let us make a total national commitment to achieve this goal.”
In the nearly five decades since, real progress has been made in the war against cancer but not enough. Cancer remains the second-leading cause of death in the United States. This year, an estimated 1.7 million new cases will be diagnosed, with nearly 610,000 people expected to die from the disease, according to the National Cancer Institute.
Fortunately, several recent cancer treatments show considerable promise. Among them is Chimeric Antigen Receptor (CAR) T cell therapy, which the American Society of Clinical Oncology recently named the “2018 Advance of the Year.” Three USC Viterbi researchers – Assistant Professor Stacey Finley, Professor Pin Wang and Assistant Professor Nick Graham – have just published a paper in “Biophysical Journal” that sheds light on how this new treatment works, information that could one day result in better cancer therapies with fewer side effects.
“We’re trying to dig into the molecular mechanisms,” said Graham, an assistant professor of chemical engineering and materials science. “By understanding how the CAR T cells work, we could try to design better ones.”
When the immune system functions normally, immune cells move around the body and look for pathogens that don’t belong and kill them. However, cancer cells can mask themselves, making it harder for the good cells, such as T cells, to kill them.
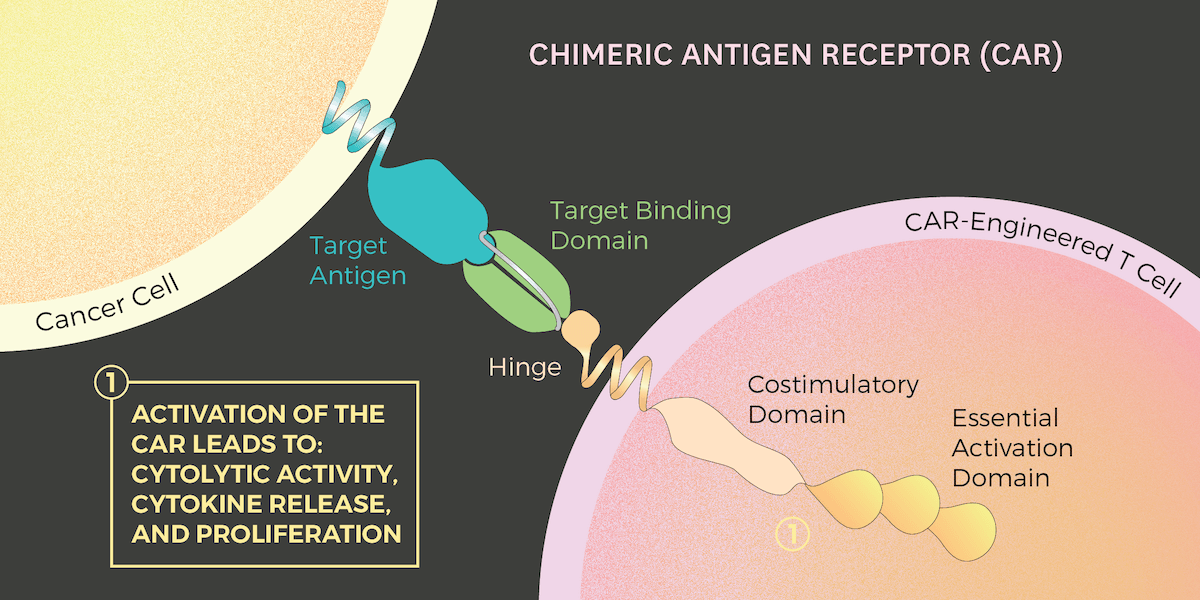
With CAR T cell therapy, a person’s T cells are removed, genetically engineered with proteins, and then injected back into the patient. The resulting CAR T cells are much better at fighting cancer cells. That’s because these modified CAR T cells have an engineered protein receptor, the CAR, that can bind to cancer cells. When this occurs, a signal from the CAR tells the T cell to begin destroying the cancer by secreting the chemicals perforin and granzyme.
CAR T cell therapies
Earlier this year, the U.S. Food and Drug Administration approved the first CAR T cell therapy for the treatment of some people with advanced leukemia and a form of lymphoma, both blood cancers. Early results have shown great promise. However, in early tests, the CAR-T cell therapies have so far proven much less effective against breast, lung, prostate and other solid-tumor cancers. Additionally, some people undergoing CAR T cell therapy have experienced significant side effects; a few have even died.

Professors Stacey Finley, Nick Graham and Pin Wang are collaborating to better understand how CAR T cells work, information that could lead to improved cancer treatments. (Image/Created by Michelle Henry)
The trio of USC researchers hope their work will greatly improve CAR T cell therapies by uncovering the complicated process by which CARs activate cancer fighting cells. Specifically, they are examining a process called phosphorylation, which is a chemical reaction that occurs when the CAR receptor bumps up against a cancer cell and sends a signal to the T cell to attack the bad cells.
“I think what’s most exciting is that we’re really adding to the field an understanding of which sites on the CAR are becoming phosphorylated, how quickly that happens and the amount of phosphorylation of each site,” said Finley, the Gordon S. Marshall Early Career Chair and assistant professor of biomedical engineering, chemical engineering and materials science and biological sciences.
Through their research, Finley, Wang and Graham have learned when and how much phosphorylation occurs on the CAR’s six sites, which, in an imperfect analogy, could be imagined as “docking hubs,” in Graham’s words. Additionally, they have found that no “gatekeeper” exists, meaning that no single CAR site must be phosphorylated before the others. Until now, scholars only had a general idea about the phosphorylation process, making it difficult to bioengineer CAR T cells that could successfully fight against complex and complicated breast, lung and other solid-tumor cancers.
Better cancer-fighting CARs
Going forward, Finley, Wang and Graham hope to leverage their findings into engineering more effective cancer-fighting CARs with fewer side effects. This could mean having phosphorylation take place quicker and more intensely at certain CAR sites, depending on the complexity of the targeted cancer cells. Alternately, the USC researchers might engineer CARs to phosphorylate less, thereby preventing the cancer-fighting T- and other cells from becoming too aggressive and killing healthy cells – a problem that has cropped up with early CAR T cell cancer treatments.
Already, Finley has built quantitative models that holds great promise.
“Once we have these tools and quantitative models, we should be able to apply them to a variety of different designs of CARs,” said Finley, whose research group has an expertise in mathematical models. “Maybe you could use a model, before you do an experiment, to see if this new design would work. Instead of having to do as many tedious experiments in the lab, you could build a predictive mathematical model to screen the best design.”
Added Wang, the Zohrab A. Kaprielian Fellow in Engineering and professor of chemical engineering and materials science, and biomedical engineering: “If you want to make the T cells more potent, the question is how best to design the CAR. That’s our research’s goal, I think.”
Published on November 1st, 2018
Last updated on November 1st, 2018