
PHOTO/Chokniti Khongchum, Pexels.
In World War I, German forces used concentrated chloropicrin, a harmful chemical compound, to deter soldiers by creating irritation and illness. In modern times, chloropicrin is used as a pesticide. It could also be a potential byproduct of certain processes used to treat wastewater.
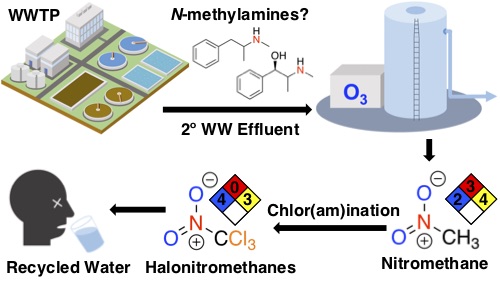
USC Viterbi researchers, including Assistant Professor in the Sonny Astani Department of Civil Engineering Daniel McCurry and Ph.D. student Jiaming Lily Shi, found that a particular wastewater treatment process, namely ozonation, could convert chemicals present in the water to chloropicrin.
Among their key findings, published this month in Environmental Science and Technology, was that wastewater reuse treatment processes that use ozonation—adding the chemical compound ozone—to disinfect drinking water create nitromethane, a chemical used in dry cleaning and as a fuel additive in drag racing. Dangerous enough on its own, when nitromethane interacts with chlorine, which is required in making treated wastewater potable, it can result in the formation of the highly toxic compound chloropicrin.
The results are predicated on the idea that nitromethane persists in the wastewater treatment process, which is likely, as no part of the current water reuse processes is currently focused on breaking down such a compound, McCurry said. A highly toxic compound, chloropicrin is currently unregulated, but has been studied as a disinfection byproduct in drinking water since the 80s with relatively little progress in identifying its precursors and formation mechanism until now.
The Chemical Precursors
As part of the study, McCurry and Shi worked to investigate potential precursors to the formation of nitromethane, which has the chemical formula CH3NO2. As it is unlikely to occur naturally in wastewater, the researchers theorized its formation comes from the addition of ozone or O3.
“We suspected that any chemical having a functional group called an N-methylamine could serve as an efficient nitromethane precursor during ozonation,” McCurry said. One such category of precursors for its formation, as reported in the study, is stimulant drugs, including ephedrine, methamphetamine and certain anti-depressants such as Prozac and Zoloft.
While ozone has been proven to be effective in removing many pharmaceutical compounds and trace contaminants from wastewater, it has been recently shown in certain circumstances to increase the toxicity of the final wastewater effluent—or the post-treatment water. Further concern results from the addition of chlorine or Cl2, which is added during final steps of treatment to make wastewater drinkable. The resulting compound–Cl3CNO2—is chloropicrin. Even at low concentrations, chloropicrin has historically demonstrated harmful effects, ranging from eye and skin irritation to asthma and vomiting.
“We found in model systems in the lab that we could convert certain drugs (e.g. ephedrine) very efficiently to nitromethane with ozone, and we couldn’t find one that didn’t form at least a little nitromethane (e.g. 8% yield from methamphetamine),” McCurry said. “We can’t yet pin the nitromethane on any particular chemical in wastewater, but what we can say for sure is that wastewater has all of these chemicals in it, and they all form nitromethane/chloropicrin.”
From wastewater treatment to potable reuse, this diagram shows one scenario for chemical treatment processes that can produce dangerous byproducts including nitromethane and Chloropicrin. IMAGE/DAN MCCURRY.
Indications for persistence of nitromethane in the water treatment cycle
McCurry and Shi are currently evaluating whether nitromethane does indeed persist in reuse treatment processes. If ozone is often the first step during water reuse treatment processes, would subsequent treatment steps do a good job of getting rid of the nitromethane formed upon its infusion? Initial results suggest that traditional treatment systems, which use reverse-osmosis and an advanced oxidation process do very little to remove or destroy nitromethane. However, an alternate treatment method, one that uses ozone followed by biologically-active carbon, seems to do a good job of clearing nitromethane.
Ultimately, as ozonation has become more common and water shortages continue expanding the need for more sustainable water practices, such as potable water reuse, it’s imperative chemical byproducts of treatment processes are more carefully studied, McCurry said.
This study was funded by the Foundation for Cross-Connection Control and Hydraulic Researchand the Rose Hills Foundation.
Published on February 3rd, 2020
Last updated on March 31st, 2022











